 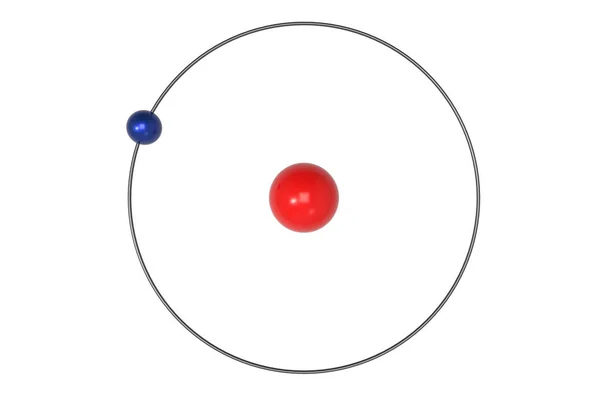
When an electron at the ground state absorbs a photon whose energy equals the difference between the ground and second state, the electron becomes excited and transitions from the ground state to the n= 2 excited state. The excited states are labeled n equal to 2, 3, 4, and so on. The lowest energy state is the ground state, which is n equal to one. The quantum number, n, is used to label the different energy states. This change can only occur if the absorbed or emitted energy equals the difference between the two states. To change energy states, an electron must move from one orbit to another by either absorbing or emitting energy. An electron can only emit energy by changing energy states. Importantly, an atom cannot discharge energy while its electrons are in stationary states. Niels Bohr proposed a model for the hydrogen atom in 1913 that described discrete energy states are associated with a fixed electron orbit around the nucleus. In this lab, you will measure and observe the emission spectrums of hydrogen, helium, and neon and use the Rydberg Equation to determine the location of the spectral lines. The initial levels marked by n-initial represent the higher energy level that the electrons excite to and n final is the lower energy level that the electrons relax back to. In this equation, lambda is the recorded wavelength, and R H is the Rydberg constant. The combination of the Balmer equation and the Bohr Model gives us the Rydberg equation, which describes the spectral lines of many different elements. Here, lambda is the observed wavelength, c is a constant, n is the lower energy level of two, and m is the higher energy level. Johann Balmer quantified the visible lines using the Balmer formula. Additional spectral lines can be measured outside of the visible range. The cyan, blue, and violet lines correspond to energy level transitions from n = 4, 5, and 6, respectively, back down to n = 2. The h alpha line is the red line at 656 nm and occurs due to the transition from n= 3 to n = 2. The visible light spectrum for the Balmer Series appears as spectral lines at 410, 434, 486, and 656 nm. The Balmer Series of spectral lines occurs when electrons transition from an energy level higher than n = 3 back down to n = 2. For example, there are six named series of spectral lines for hydrogen, one of which is the Balmer Series. Since different atoms have different energy levels, these spectral lines vary from element to element and depend on the transitions those electrons make between energy states when excited. With pure elemental species, the emission behavior appears as lines of specific wavelengths rather than a broad spectrum. The emission spectrum is a measure of emitted radiation across a range of wavelengths. High energy emitted light results from electrons relaxing from a higher energy level, and low energy emitted light results from electrons relaxing from a lower energy level. The wavelength of the emitted light depends on the difference between the high and low energy levels. These shells are the same as the energy levels and are labeled by n. You may be familiar with these energy states, as they are depicted in the Bohr Model, which describes an atom as a nucleus with orbiting electrons in shells or orbitals. When the electrons relax down to either a lower energy state or to the ground state, the excess energy is released as emitted light. However, when an atom absorbs energy, the electrons become excited and move to a higher energy level. 
The electrons in an atom traditionally exist at their lowest energy state, called the ground state, and labeled as n equal to one. multiple of $ \frac =0.Atoms and molecules exhibit very interesting behavior when they absorb and release energy. Only these orbits are permitted for electron which angular momentum (L = m vr) is whole no. or K, L, M ….These integral numbers are known as Principal Quantum numbers In an atom electron revolve around the nucleus in certain definite circular paths known as orbit or energy shells.Įach orbit is associated with definite energy hence known as energy levels or shells. He used Planck's concept of quantisation of energy. Neils Bohr (1913) was the first to explain quantitatively the general features of the structure of hydrogen atom and its spectrum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
